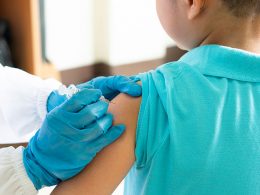
The National Institutes of Health (NIH) is putting together a study to find out why people are showing severe allergic reactions when administered with the vaccine made by Pfizer-BioNTech, although such cases are rare, the Hill reports.
The study aims to discover what component of the vaccine causes people to undergo anaphylaxis, a severe allergic reaction to the vaccine, which can prove life-threatening in many cases. Although a very challenging effort, scientists would be hoping to achieve their goal in the weeks ahead as more and more people in the country get vaccinated.
For this study, the NIH is seeking participants who have a history of severe anaphylaxis and would receive the vaccine under close medical supervision. Daniel Rotrosen, director of the Division of Allergy, Immunology, and Transplantation at the National Institute of Allergy and Infectious Diseases, said that it was not a simple study design, as the study requires highly-allergic individuals, which would not be easy to find.
While both Pfizer and Moderna vaccines underwent rigorous scientific scrutiny by various independent panels of experts, no cases of anaphylaxis have happened with the newly-approved vaccines by Moderna, which would be administered to the public this week.
The government has urged the public not to be discouraged by the negligible adverse reactions that occur from vaccination. They recommend that people who get vaccinated should remain within a clinic for up to 30 minutes if they have a history of severe allergic reactions. However, using epinephrine, anaphylaxis can be quickly arrested.
According to the US Centers for Disease Control and Prevention (CDC), as of Sunday afternoon, about 556,000 people have been vaccinated in the United States. On the other hand, as of Saturday, the CDC recorded six cases of anaphylaxis in people who received the vaccine made by Pfizer-BioNTech, the Washington Post writes.
While the vaccines do not contain the coronavirus itself, it has a synthetic messenger RNA that instructs cells to replicate a protein of the coronavirus. The immune system subsequently builds up antibodies to fight off the infections by the real virus. Following vaccinations, people can experience mild symptoms such as headaches, fatigue, or pain at the injection site. These symptoms disappear within a day or two.
According to Peter Marks, a director of the US Food and Drug Administration (FDA), polyethylene glycol is considered a prime suspect for the negative reactions experienced after vaccination. PEG, as the compound is known is found in other drugs and cosmetics.
Retrosen said the intended study by the NIH would involve hundreds of participants who would be stationed at various clinics across the country. He said the volunteers would be under strict observations and follow-up to evaluate their reactions.
Professor N. Franklin Adkinson of the Johns Hopkins Medical School re-echoed the same sentiments as Marks, identifying PEG as a leading suspect in anaphylaxis reactions caused by the vaccine.
The authorities commenced looking into cases involving anaphylaxis when a middle-aged woman and an employee of Bartlett Regional Hospital in Juneau, Alaska, suffered multiple phases of anaphylaxis after vaccination. She had to be administered epinephrine and other medications before she recovered.
Vaccination in the US is ongoing and several government officials have been publicly vaccinated to build up the public’s confidence in the inoculation program.
Source: thehill.com